The COVID vaccine and bivalent booster for kids 6 months-5 years
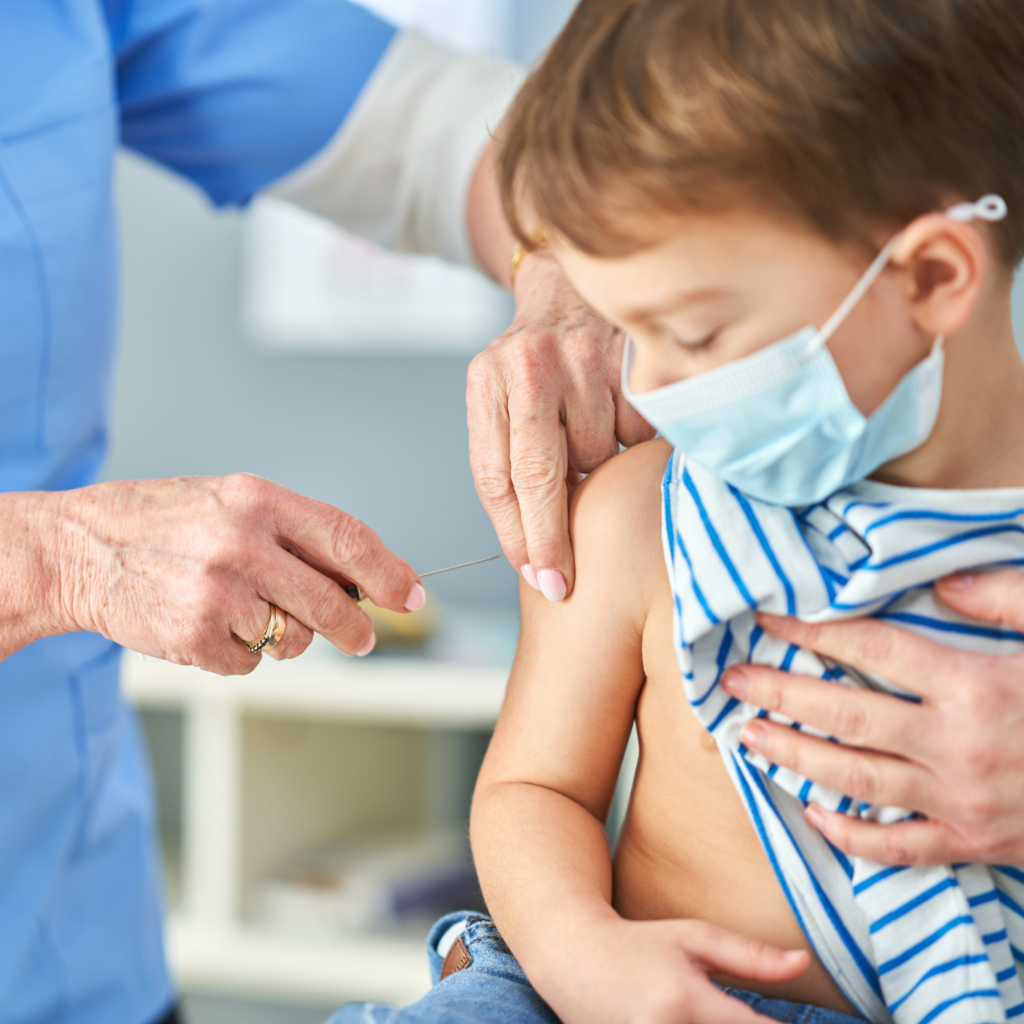
Curious about the COVID vaccine for kids 6 months-5 years? This blog is for you. Fair warning: it’s long. But, I think it really includes important summaries and visuals to help guide your decision.
Update: March 22, 2023
Last week, the U.S. Food and Drug Administration (FDA) released a press statement with an important COVID-19 update. The FDA authorized emergency use authorization (EUA) of the Pfizer-BioNTech bivalent COVID-19 vaccine as a booster. The omicron BA.4/BA.5 adapted bivalent COVID-19 vaccine was approved as a booster for children ages 6 months through 4 years, depending on their eligibility.
Who can get the Pfizer-BioNTech bivalent COVID-19 vaccine?
There are several criteria for eligibility to receive the bivalent COVID-19 vaccine. This authorization is only for children ages 6 months to 4 years, commonly referred to as children under 5 years. These children are eligible if they previously completed the three-dose vaccine series with the Pfizer monovalent COVID-19 vaccine more than 2 months ago. The bivalent vaccine is already authorized for children under 5 years as the third dose in the 3-dose series. This has been a recommendation since December 2022 – children receive the first two doses of the Pfizer monovalent COVID-19 vaccine, and the third dose is the Pfizer bivalent COVID-19 vaccine to complete the 3-dose series.
Of note – if children completed the 3-dose series with the Pfizer bivalent COVID-19 vaccine as the third dose, they are not eligible for a booster. This vaccine series is considered to provide adequate protection against severe COVID-19 complications, such as hospitalization and death.
A quick recap – children 6 months through 4 years are now eligible for a bivalent booster if they completed the Pfizer 3-dose series with the monovalent vaccine two months ago. Children 6 months through 4 years are already authorized to get the bivalent vaccine if they are due for the third dose in the 3-dose series.
A few details about the Bivalent vaccine
The bivalent vaccine is different from the monovalent vaccine in the protection offered. The bivalent vaccine offers protection against the original COVID-19 strain and the omicron BA.4/BA.5 strain.
Specifically, the bivalent vaccine offers better protection against COVID-19 caused by the omicron variant.
What we know about the Pfizer-BioNTech bivalent COVID-19 vaccine
There is an ongoing study, Phase 1/2/3 (NCT05543616), evaluating the efficacy and safety of the COVID-19 vaccine in healthy children and young adults. Data from this study was used to assess the bivalent vaccine.
The FDA assessed a sample of 60 children ages 6 months through 4 years. These children completed the 3-dose vaccine series with the monovalent vaccine and then received the bivalent booster. One month after the bivalent vaccine, the children demonstrated an adequate immune response to the original COVID-19 strain and the omicron BA.4/BA.5 strain.
The FDA evaluated the safety based on prior clinical studies. These children completed the 3-dose vaccine series with the monovalent vaccine and then received the bivalent booster. The clinical study broke down the safety of the bivalent vaccine for several age groups. The study revealed that the most common side effects for children ages 6 months through 23 months were irritability, drowsiness, injection site redness, pain and swelling, decreased appetite, fatigue, and fever. The most common side effects for children ages 2 to 4 years included fatigue, injection site pain, redness and swelling, diarrhea, vomiting, headache, joint pain, and chills. Lastly, children 5 to 11 years most commonly reported fatigue, headache, muscle pain, joint pain, chills, fever, vomiting, diarrhea, injection site pain, swelling and redness, and swelling of the lymph nodes in the same arm of the injection.
What are the plans for the future?
Currently, the U.S. FDA authorized the EUA of the Pfizer-BioNTech bivalent COVID-19 vaccine as a booster. This means that the emergency use of the bivalent booster is only authorized for the duration of the specified circumstances.
In the European Union (EU), the bivalent booster is authorized for children ages 5 years and older. Pfizer and BioNTech submitted an application to the European Medicines Agency to extend the bivalent vaccine authorization to include use for children 6 months through 4 years as both the primary series and a booster.
Pfizer and BioNTech plan to submit applications to regulatory agencies worldwide for the use of the omicron BA.4/BA.5 adapted bivalent COVID-19 vaccine for children under 5 years.
How to decide if your child is a good fit for the vaccine
This is an important conversation to have with your child’s pediatrician. There are several specific criteria for eligibility and they will be able to guide you through if the vaccine is a good fit. It’s important to consider and discuss the timing of the vaccine. In particular, if your child has recently had COVID since it’s commonly accepted that this offers protection against severe COVID-19 illness.
You can consider risks vs. benefits. Fact sheets are required for vaccines – commonly, these outline the indications for use, important safety information, common side effects, and when to seek medical attention. If you have questions or concerns while reviewing the information, you can further discuss these with your child’s pediatrician prior to consenting to the vaccine.
***As of December 10th, 1.64 Million children between 6 months to 5 years have received at least one dose of the vaccine. You can see updated vaccination numbers here. *** Clinically, we are seeing the typical side effects reported in the trials and no concerns.
Update: December 15, 2022
The bivalent booster was rolled out in September and expanded to children as young as 5 in October. Now, it’s authorized for children as young as 6 months old.
What does bivalent even mean?
Bivalent means the vaccine offers protection against two different types (strains/variants) of COVID—the original strain and the Omicron BA4/BA5 strain. Monovalent is how we discuss the original COVID vaccine all of us who got the COVID vaccine received. Fun fact: The flu vaccine is a quadrivalent vaccine this year so 4 strains. Thought the terminology would be helpful before we continue.
So, when it comes to kiddos; here was the OLD recommendation:
- Moderna: For kids 6 months-4 years-Two doses:4-8 weeks apart
- Pfizer: For kids 6 months-4 years-A three dose series with Dose 2 that is 3-8 weeks after Dose 1 and Dose 3 that is 8 weeks from Dose 2.
Now, the bivalent booster is here and the recommendations are such:
- Moderna: Children 6 months through 5 complete the two-dose series AND then receive a bivalent booster 2 months after completing the series (so a total of three injections).
- Dose: 10 micrograms (monovalent is 25 micrograms).
- Composition: Half with original formulation and half specific to BA4/BA5 variants.
- Pfizer: Children 6 months through 4 complete Dose 1 and Dose 2 would be the original vaccines in the series, but now DOSE 3 would be the NEW bivalent vaccine. (also, a total of three injections like the Moderna vaccine). An important thing to remember: If your child has already completed the three-dose Pfizer series, they are not eligible for the bivalent booster. This is because, there is not data to determine if the kids who received the original Pfizer series actually NEED a booster, so more information will be reviewed early next year if those who completed the 3-dose Pfizer series even need a 4th vaccine which is the bivalent booster. I am not annoyed by this and think it’s doing due diligence before recommending something that MAY not be needed. Updated guidance IS down the pipeline.
- Dose: 3 micrograms (monovalent is the same)
- Composition: Half with original formulation and half specific to BA4/BA5 variants.
In both Moderna and Pfizer, monovalent vaccines are given as the first and second doses (monovalent was the original COVID vaccine) and bivalent vaccines cannot be given for these doses. Now for common questions:
How do they know this is safe if it wasn’t studied directly on kids this age?
There is safety date from older groups and also safety data from the monovalent. Think of this like the flu vaccine. The flu vaccine gets updated every season to account for new variants. The science behind the vaccine isn’t changing, just the composition of variants it’s geared too. Billions of doses of the COVID-19 vaccine have been given. Here is more about the safety monitoring of the bivalent vaccine.
Were the bivalent vaccines effective?
The real-world findings come following updated clinical trial data from both Pfizer-BioNTech and Moderna on their bivalent boosters earlier in November. Both companies’ bivalent boosters produced significantly higher immune responses, compared to the original COVID-19 vaccines, with similar safety and tolerability profiles. As an example, Pfizer’s data showed this: One-month after a 30-µg booster dose of the bivalent vaccine, Omicron BA.4/BA.5-neutralizing antibody titers increased 13.2-fold from pre-booster levels in adults older than 55 years of age and 9.5-fold in adults 18 to 55 years of age, compared to a 2.9-fold increase in adults older than 55 years or age who received the original booster vaccine. So, there IS efficacy in adults and this will likely extrapolate to kiddos too.
Would you recommend this?
Yes, I would. These bivalent vaccines are more geared towards Omicron variants. Although it’s not a 100% match, it’s closer than the original series so I believe this update to be great and I wouldn’t be hesitant to get the bivalent. That being said, I don’t know full efficacy (nobody does) given it’s new but the benefits outweigh risk. See my blog for more questions and things to consider about COVID vaccination. Ryaan has had COVID twice and when we were set to get him the COVID vaccine series he was sick with a fever, so we had to push back. One week later, the bivalent booster came out for adults. My husband and I decided to wait for the booster to be authorized for Ryaan’s age group and then get him started. We’re happy because this means he will get the Pfizer vaccine with the updated booster now as the third dose. So, we are set to start the series in January. I like that it’s more geared towards Omicron from a higher efficacy standpoint. Also, I am seeing COVID in kids creep back up in the past two weeks and many of them look like they have flu-like symptoms. Many do make full recoveries, but I’d like to add more risk-reduction with updated vaccines if we can.
Can we give this vaccine with other vaccines?
Yes. As the science is not different, there is no concern with co-administration. If you prefer to keep separate you can, but don’t avoid or push-off to long or forget.
BA.4 and BA.5 are not the predominant variants, so what’s the point?
It is true that BQ1 and BQ1.1 are the dominant variants, but these are also Omicron subvariants. The monovalent vaccines had no protection against Omicron variants. So, these boosters are expected to provide more protection against these new circulating Omicron variants but how much is too soon to tell. I always like to be transparent with scientific data, so yes nobody knows how effective this booster is to current subvariants, but based on the way variants work; it’s likely MORE than the original monovalent vaccine.
If you haven’t gotten your child the COVID vaccine yet, this is a better time to consider. The vaccines are becoming more specific to what’s circulating so benefit is more there than before. The degree of efficacy is TBD, but safety profile remains consistent.
If you are searching for vaccines, visit vaccines.gov or ask your Pediatrician’s office. It may take some offices a few weeks to get their supply or determine if they will carry it. Any Pediatrician who is currently carrying the vaccine for children 6 months-5 years should like get the updated booster.
Update from June 2022
The Vaccines and Related Biological Products Advisory Committee (VRBPAC) met on Wednesday, June 15 and voted unanimously 21-0 to authorize the Pfizer vaccine for children 6 months to 4 years and the Moderna vaccine for children 6 months-5 years.
On June 17th and 18th, the CDC Advisory Committee on Immunization Practices (ACIP) met to also discuss the data, logistics, administration and so much more. They had an unanimous 12-0 vote to authorize both vaccines. They made it clear they will vote on each vaccine compared to NO vaccine and not be comparing the two given comparison is not completely equal (both studies were not done at the exact same time, had different numbers of participants, different dosing, small sample sizes for looking at symptomatic prevention, and so much more).
For the ACIP meeting presentation from 6/17/22, check it out here. For their meeting presentation from 6/18/22, check it out here. I found these slides to be more digestible than the FDA slides with more practical guidance.
I am so grateful to have been able to listen in on all of the meetings. My schedule allowed me to do this and it was fascinating being able to learn and listen in.
When deciding to issue the EUA, both committees asked their members the question: Why would an EUA be needed? Does the benefit outweigh the risks to issue an EUA?
When deciding to issue an EUA, the committees look at disease burden (deaths, hospitalizations, burden to healthcare systems, and burden to working parents having to leave the work force).
So what was the disease burden for COVID-19 and kids in this age group?
This age group can get COVID-19:
- Total # of cases under 5 between March 1, 2020-June 7, 2022
- <1: 567,535
- 1-4: 1,913,969
Increased hospitalization visits, especially during Omicron.
This is a burden to healthcare resources where during the Omicron surge, close to 15% of all ER visits were due to Omicron. We don’t know if that was just for testing or if the child was really sick warranting an ER visit, but this is a high percentage of visits to an ER (hospital bills/time/staff/etc) so COVID-19 is considered a health system burden even for this age group:
Need for ICU and oxygen support for this age group.
In general, 0.8% of all children in this age group who tested positive for COVID needed to be hospitalized. By percentage, this is low, but it still resulted in a high volume of children in the hospital given the total number of cases. Some parents will hear 0.8% and think thats nothing whereas others will realize thats about 20,000 children being hospitalized and want more protection–that is why a vaccine is available based on your level of comfort of risk.
Although total hospitalizations in this age group during the Omicron surge was low compared to total number of cases (535 children 6 months-4 years), 23.6% needed ICU admission, 16.2% high flow nasal cannula, 6.4% needed BiPAP or CPAP (advanced oxygen support), and 6.4% needed intubation. Overall risk is LOW for hospitalization of this age group compared to the large number of kids who had Omicron, however we did see some over ¼ need ICU care during Omicron. One comment was what about the current variant that is not causing as much hospitalizations. Could things be improving? We don’t know how future variants will behave, hence why vaccination can help.
Hospitalizations of COVID-19 in kids as it compares to other common childhood vaccine-preventable illnesses.
Before the creation of the Hep A vaccine, varicella, and pneumococcal vaccines, hospitalizations for COVID-19 were comparable to varicella and pneumococcal hospitalization. This shows disease burden is similar to those illnesses which we do give routine vaccines for now and have resulted in less hospitalizations.
COVID-19 Deaths in this age group
Between January 1, 2020 – May 11, 2022 were 202 children (we do not know of the underlying health status of these children, but these are 202 lives lost that we hope to prevent with vaccination). Similar to hospitalizations, some parents will hear 202 deaths and think thats a low percentage compared to total number of cases whereas other parents will see that 202 children in this age group did die from this virus and that’s 202 too many. Vaccines are created against viruses/illnesses that have caused deaths or complications, hence why an EUA makes sense to allow parents to add more protection and risk-reduction if they wish to.
Deaths related to COVID-19 in this age group as it compares to other vaccine-preventable illnesses.
Similar to the graph shown above with hospitalization burden, they presented death data as it compares to other vaccine-preventable illnesses pre-creation of vaccines. The disease burden for COVID-19 is high in this age group.
Looking at all of this data above, we can see that although overall risk of hospitalization and death is low (hospitalization rate was 0.8%- could be lower given under-reporting of cases), there is 100% a disease burden that would warrant having a safe vaccine available for children of this age group.
Now let’s get into each vaccine (Moderna and Pfizer)
Moderna is proposing a 2-dose series of 25mcg each while Pfizer is proposing a 3-dose series of 3mcg each. (Thank you @sciencewhizliz for this graphic).
Below is the dosing regimen as outlined from CDC’s ACIP committee (of note, as of December 10th, 2022, this recommendation has changed to include the updated bivalent booster):
ACIP is allowing a 3-8 week interval between dose 1 and 2 of Pfizer and a 4-8 week interval of dose 1 and 2 of Moderna for healthy children.
A shorter interval (3 or 4 weeks vs the 8 weeks) would be recommended if the child is immunocompromised, high risk for severe disease, living with someone who is immunocompromised, or if there is high community transmission).
Longer spacing intervals could reduce the risk of myocarditis and some studies showing it MAY optimize vaccine effectiveness. Of note, we have not heard of any myocarditis cases in this age group thus far!
Speak to your clinician on their recommendation for spacing. I am recommending 3-4 weeks between the first and second dose given the presence of new variants.
How is efficacy determined in pediatric vaccine studies?
In terms of efficacy for both vaccines, when evaluating pediatric vaccines they rely heavily on data from older children/adults. This is called an immunobridging study. They infer vaccine effectiveness by comparing the antibody response in this age group as it compares to antibody responses in adults. In adult trials, they look at efficacy against symptomatic illness and although we did get some data on that in the trials, adequate antibody data is required to infer likely immune response. This makes sense because pediatric trials tend to be a smaller number of subjects in the study and true effectiveness can be hard to determine if not many kids get a COVID infection in the trial (which we did see in both trials, especially in Pfizer).
Antibodies are essential for neutralizing the virus on entry and preventing infection, and they can easily be measured in a drop or two of blood.
Both committees (FDA and CDC) stressed that immunobridging studies are more important than “symptomatic efficacy” given low sample sizes (remember this as you read on).
So, comparing the two and saying one is BY FAR superior is not fair and both are adequate for immune antibody response compared to older children or adults when the series is completed as scheduled.
Let’s talk about the Moderna Vaccine
If you would like to see their data/presentation, check it out here.
- Timing: December 2021 to February 2022 (Omicron surge)
- Total Participants:
- 2,355 children 6-23 months
- 4,048 children 2-5
- 2/3 received the vaccine and 1/3 received a saline placebo
- Dosing: Dosing of this vaccine was set at 25mcg 4 weeks apart for children 6 months-5 years. They considered 25mcg and 50mcg (as a reminder, adults received 100mcg of the Moderna vaccine). Once they saw response with the 25mcg, they did not need to go up to 50mcg.
- Follow-up time post dose 2: 2.5 months
- Demographics: Racial demographics: white 76%, 11% multiracial, 5% black, 6% Asian
- Comorbidities: 421 kids 2-5 years had Asthma, Obesity, Cardiac disorder- Includes Aortic dilatation, Aortic dissection, Arterial switch operation, Atrioventricular block first degree, Bicuspid aortic valve, Cardiac ablation, Cardiac operation, Coarctation of the aorta, Double outlet right ventricle, Extrasystoles, Fallot tetralogy, Heart block congenital, Heart disease congenital, Hypertrophic cardiomyopathy, Hypoplastic left heart syndrome, Palpitations, Patent ductus arteriosus, Pulmonary valve disease, Pulmonary valve stenosis, Supraventricular tachycardia, Systemic-pulmonary artery shunt, Transposition of the great vessels.
- Side Effect Profile: The most common side effect was pain (61% after dose 1 and 71% after dose two. This makes sense given pain is a common side effect of most vaccines. The report of pain was likely higher in this age group given the higher dose of 25mcg as compared to data in the Pfizer trial).
- For children 6 months-36 months: Common side effects included: fever, irritability, crying, sleepiness, loss of appetite
- For children 37 months-5 years: Common side effects included fever, headache, myalgia, arthralgia, nausea vomiting.
For most children, symptoms resolved within 48 hours. A small percentage of children did have fever for 4-5 days. Fever was more common in children under 2 years of age.
There were NO deaths, myocarditis, or pericarditis.
Of note, there was one child who did experience a febrile seizure after the vaccine including a rash. They did not have any neurological complications or history of febrile seizure and resolved as febrile seizures do. Of note, febrile seizures are common in 3-5% of the pediatric population.
Effectiveness against symptomatic COVID:
Remember that immunobridging studies can infer efficacy. Limitations to the data below for preventing symptomatic COVID include smaller sample sizes as compared to adults. We will get even MORE data once more children get vaccinated.
Immunobridging: antibody levels measured 28 days after the second dose for participants without prior infection. This was compared to individuals who received the vaccine ages 18-25.
Below is symptomatic efficacy data, which is different than the antibody response (“immunobridging”) needed to be authorized.
6 months-23 months old
Efficacy 50.6% based on the CDC definition of COVID
24 months-<5 years old
Efficacy 36.8% based on the CDC definition of COVID
These numbers may seem low for symptomatic COVID, however it is comparable to real world data for adults, the COVID vaccine, and Omicron
On the left, we look at Kaiser Permanente data shows 44% efficacy against Omicron in adults and children were 36.8% in children and 50.6% so this is comparable to real-world data and we know a booster will likely be needed down the line. So when considering efficacy, it is comparable to the adult population.
More about the Moderna Vaccine:
- From the ACIP meeting, they discussed that 6-8% of children had prior COVID in the study.
Seropositive means a child was positive for COVID. If you notice on the left for 2-5 year olds, vaccination provided a 178-fold increase after NO prior COVID infection whereas a 37-fold increase after a past COVID infection. This shows that there was a stronger antibody response for those who did not have COVID before but there WAS a response also for those who did have COVID, showing the benefit for vaccination for those with prior illness.
- They did not have any data for babies who were born premature and are currently conducting a study for premature babies.
- No children received the vaccine co-administered with other childhood vaccines. They did allow the flu vaccine to be offset by two weeks and other vaccines offset by four weeks. But we do not have any information on co-admin
- Booster: Moderna should have a booster by early fall. They will be reviewing the data and likely interval would be 5 months (congruent to adults). They did mention it’s hard to understand if a booster is needed because of waning immunity or Omicron or future variants similar to adults. From a recent press release, Moderna did mention that the booster they are studying for adults can protect against current variants. The antibody response was lower than for current variants, but this same vaccine may be modified in dosing for this age population as well.
Now let’s talk about Pfizer
If you would like to see their data/presentation, check it out here.
- Timing: June 2021 to April 2022 (Delta and Omicron surge). It was longer because they started with a 3mcg two-dose series which did not meet necessary data so they ended up needing to expand to a 3-dose series which coincided with the Omicron surge. (The three-part series met immunobridging data needed to be authorized).
- Total Participants:
- 1,776 children 6-23 months
- 2.750 children 2-5
- 2/3 received the vaccine and 1/3 received a saline placebo
- Dosing: 3 doses of 3mcg (with intervals at 3 weeks and 8 weeks) for children 6 months-4 years.
- Follow-up time post dose 2: 1.3 months
- Demographics: White 76%, 11% multiracial, 5% black, 6% Asian
- Comorbidities: Among participants 6-23 months of age, 4.2% of vaccine recipients and 5.7% of placebo recipients had a comorbidity at baseline. The most common was prematurity (1.6%), followed by asthma and congenital heart disease (each 0.8%), cardiovascular disease (0.4%), neurologic disorder (0.3%), chronic lung disease (0.2%). Congenital heart disease was more frequent in the placebo group (1.8%) than the vaccine group (0.8%).
- Among participants 2-4 years of age, 12.1% of vaccine recipients and 14.2% of placebo recipients had a comorbidity (including obesity) at baseline. Obesity was reported in 6.5% of vaccine and 4.9% of placebo recipients. Other than obesity, the most common comorbidities at baseline in BNT162b2 group included asthma (2.8%), prematurity (1.1%), congenital heart disease (0.7%) and neurologic disorders (0.6%). One vaccine participant had a baseline comorbidity of immunocompromised condition (neutropenia).
- Side Effect Profile: Lower side effect profile than Moderna, but neither trial had any serious issues and all common side effects were self-limited and resolved usually within 24 hours. The Moderna vaccine likely had more common side effects of pain given the dose size, but was safe in trials.
For example, pain at site was reported at 30% for Pfizer versus 61-71% for Moderna.
There were NO deaths, anaphylaxis, myocarditis, bell’s palsy, MIS-C
Of note, there were five children who did experience a febrile seizure however it was 21 days-68 days post-vaccine so was determined to be UNRELATED to the vaccine (which I agree since temporally is so far). Of note, febrile seizures are common in 3-5% of the pediatric population.
Effectiveness against symptomatic COVID:
Antibody levels measured one month after the second dose for participants without prior infection. This was compared to individuals who received the vaccine ages 18-25.
For 2-5 year olds, The vaccine was not effective after TWO doses as it did not meet immunobridging criteria but DID after three doses (for both groups). This is likely due to the dose of 3mcg that met immunobridging criteria for younger children (6 months-2 years), but not for this age group. For both age groups, antibody response was met after finishing the 3-dose series. So, if your child gets two doses of Pfizer per their antibody comparison to adults/elders, they are not protected in comparative levels UNTIL two weeks after dose three.
Below is symptomatic efficacy data, which is different than the antibody response (“immunobridging”) needed to be authorized. Remember, similar to Moderna and per the ACIP. we will get better data about this as we study larger population data.
Looking at far left, the vaccine was 21.8% effective against symptomatic COVID after 2 doses and 80.3% after the third dose in children 6 months to <5 years.
For children 6 months to <2 years, it was 4.2% effective after 2 doses and 75.5% after dose 3. For children 2-5 years, it was 82.3% effective after dose 3.
This is important to remember as effectiveness is low after two doses (which we knew back in the Winter which pushed back the EUA). This is likely due to a low dose of 3mcg.
The extremely small samples mean we cannot conclude much of anything about efficacy against confirmed illness. In six-to-23-month group there were three total cases—1 in the vaccine recipients and 2 in the placebo group). For children 2-5, there were 7 total COVID cases (2 in the vaccine group and 5 in the placebo group).
31% of total trial participants received a third dose (992 received the third dose and 464 got a 3rd dose of placebo saline). So, the sample size is small.
So, we are relying heavily on the antibody response (immunobridging data comparing to older people) after three doses which met FDA criteria for authorization and is deemed effective more on that regard. This was stressed multiple times by the ACIP committee on June 18th.
More about the Pfizer Vaccine:
- From the ACIP meeting, they discussed children had prior COVID and did have an antibody response (do not have graph data).
- 1.6% of patients were premature.
- No children received the vaccine co-administered with other childhood vaccines. They did allow non-live vaccines to be given within two weeks and LIVE vaccines to be given within four weeks (LIVE vaccines are rotavirus, varicella, MMR).
Comparing both of them
BOTH vaccines met antibody thresholds needed to show efficacy compared to adults and both are authorized. Take the vaccine that is available to you. If you have a choice, you can use the following to decide:
- More trial participants: Moderna
- More real-world shots given in pediatrics: Pfizer (has given 8.4 million vaccines for 5-11 and 15 million vaccines for kids 12-17). Moderna only has given vaccines to 18+. So, we have more safety data for Pfizer for children.
- More common side effects: Moderna (likely related to higher dose, although these resolved)
- Irritability:
- Pfizer: 43.8%-51.2%
- Moderna: 54%-56%
- Decreased Appetite:
- Pfizer: 20-22%
- Moderna: 24-31% for younger kids 61-74% for 2-5
- Pain:
- Pfizer: 26.7%-30%
- Moderna: 37%-46%
- Fatigue:
- Pfizer: 24.5%-29.7%
- Moderna: 40-48%
- Irritability:
- Lower dose: Pfizer. A lower dose still met antibiody response after three doses. A lower dose can mean less of the common side effects, but do remember even those children with the 25mcg had common side effects that resolved.
- Safety for extreme adverse effects: Both (no deaths, myocarditis).
- Safety in general: Remember that 8,000 children were in these studies and 600 million mRNA vaccines have been given in the U.S. alone with continued safety profiles. Like any “medicine,” vaccines may carry low risk, but population data and the trial show overall very favorable outcomes as compared to the risk of severe illness.
- Faster protection: Moderna (but will likely need a Booster like adults). Moderna would likely work “faster” but final outcomes at end of each series are likely the same given antibody reponses. If your child has NOT had COVID and is 6 months-2 years, either vaccine would be okay (as both met immunobridging criteria–antibody response). If your child has not had COVID and is 2-5, antibody levels were not adequate after two doses of the Pfizer vaccine but were after three doses. This may make Moderna a better option for you if you want “quicker” protection (if you have the choice). Getting two doses of the Pfizer vaccine in this age group may not provide as much protection as two doses of the Moderna vaccine given the antibody levels criteria for efficacy. If your child has had COVID, you can opt for either vaccine and complete the full series. Remember, efficacy levels are truly hard to interpret with new variant circulating.
- Of note, from a recent press release, Moderna did mention that the booster they are studying for adults can protect against current variants (BA.4 and BA.5). The antibody response was lower than for current variants, but this same vaccine may be modified in dosing for this age population as well. The reality remains that this booster will become available AFTER the variant leaves–they’re saying it would be available in August, but with this variant circulating now in June and July–vaccines are playing catchup with a mutating virus. So, it’s unknown which booster children would get in the fall and how far it will be recommended from their original series.
- It’s unknown if children who receive the 3-dose Pfizer series will need a fourth dose for protection. Moderna recipients in this age group will likely need this booster being created by the fall.
Remember, your Pediatrician may not have options for both and will likely only carry one due to storage needs and cost. Take the vaccine that is available to you. If you have a choice, you could use the above-mentioned to decide which one you want based on your situation.
If that wasn’t enough information, below is what was sent to my newsletter. It’s a Q&A with common questions I’ve been getting regarding the vaccine. The below portion is more opinion and nuanced than what you’ve already read.
To sign up for my newsletter so you don’t miss the latest and greatest from me, sign up here.
How soon can my child get the vaccine?
This will depend on when your clinic/health department placed an order. Some facilities were waiting until the FDA authorization happened on Friday before they ordered. You can also check here for vaccination sites. It’s already populating many locations (even for you Florida fams!). If you have access, you can get it whenever you want.
If my child has had COVID, do we have to wait a certain amount of time to vaccinate?
The recommendation from ACIP (CDC) is that you can wait 3 months after a known COVID infection as this may result in an improved vaccine response. If your child is high risk for complications or immunocompromised, speak to your clinician if they need to wait the 3 months. The three month rule is a “may” and not an absolute. The idea is natural infection can give you 2-3 months of protection, but this is not for certain in the pediatric world and with new variants–so you could vaccinate as soon as recovered from COVID and no more than three months post-COVID. (Given the current state of variants and reinfection, I recommend vaccinating as soon as recovered or within 6 weeks of a past COVID infection).
Is it worth the (minimal, I know) risk, given kids generally do not get as sick?
The risk is very low with the vaccines based on safety studies in the trial and in the general population. In this age group, there have been 2 million cases, 20,000 hospitalizations, and 200 deaths since the pandemic began. If your child has not had COVID, there is a benefit to getting vaccinated to likely reduce this risk (what we have seen in older ages).
If your child has had COVID, vaccination immunity can help with future variants. We don’t know if the variants will continue to become less severe or if they will morph into something else; so vaccination can provide benefit. So deciding not to vaccinate is basically you feeling comfortable that the virus will not morph into something more severe (which is hard to predict).
Is the vaccine recommended even if my child has had COVID before?
Yes. The ACIP meeting particularly discussed that vaccine immunity is likely longer lasting than natural immunity. What we do not know is how the vaccine will act towards future variants (will it reduce hospitalizations, etc) but it likely will given the data we have from the Omicron surge for kids over 5+.
Is natural immunity enough to prevent against variants?
Likely no based on this study. Children over 5 who were vaccinated against COVID demonstrated higher titers against various variants than those who weren’t vaccinated. These findings suggest that antibodies produced by prior SARS-Cov-2 infection (pre-Omicron) may not neutralize the current variants, but vaccination can. This builds on evidence among adults that previous infection provides poorer protection from infection with Omicron (and likely future variants). Do remember, we do not have data of this from the current surge we are in.
If my child had COVID and it was mild, is it safe to say every future infection will be the same?
My hope is yes, but the unfortunate reality is this virus is constantly changing and surprising us and unvaccinated individuals will always be the most vulnerable to worsening potential variants. Vaccination will provide more protection than natural immunity and protect against the risk of future worsening variants IF that were to happen.
Can my vaccinated child still get a COVID infection?
Yes. Similar to adults, vaccinations will not always prevent against symptomatic COVID, BUT it can reduce risk. IF a child were to get COVID even after vaccination, their chance of hospitalization or other COVID-related issues like MIS-C is lower. So, the benefit is there for reducing hospitalizations or complications. Coronavirus is a respiratory virus so similar to flu, RSV, and influenza, reinfection is possible but severity of illness can be greatly benefited by vaccines.
Do we know if vaccination reduces the risk of hospitalization?
We will need more population data like what we saw in children 5+ here. From the 5+ year olds, we see that unvaccinated children were 2x as likely to be hospitalized than vaccinated children during the Omicron surge. In adults, we know this as well: vaccinations have helped reduce hospitalizations. We do not have data from the BA.4 surge. It is likely we will see the same with this age group, but to be completely transparent we can’t predict it because we don’t know what future variants await us (if any) and if they will evade vaccine immunity.
My kid is healthy, why do they need it?
I hear you. I have seen a lot of healthy children get COVID and do really well, but we also see some get hospitalized and some who have died. 26% of all children of this age group admitted to hospitals during the Omicron surge had no underlying medical conditions. Most made full recoveries, but if we can prevent a hospital stay for you (or worse), I would love to provide you that. And vaccines can help do that.
Any reason NOT to vaccinate?
If a child had a prior reaction to a COVID vaccine, otherwise the vaccine is recommended for all kids. I do know some parents who are on the fence because their child has had it once or twice and fared really well. Their comment: “what’s the point?” I hear you and I respect this thought process. I will say, we do not know what future variants will do (if anything) so choosing to vaccinate can add a layer of protection against future variants.
Which is better: Moderna or Pfizer?
Both met immunobridging or antibody response data needed comparable to adults. So to say one is better is difficult with the symptomatic COVID data we have. The blog above had a breakdown of each.
Take the vaccine that is available to you. If you have a choice, it depends on many factors. If your child has never had COVID, Moderna may make the most sense given better immunity quicker. If your child has had COVID, you can opt for Pfizer. Pfizer has a lower dose which likely means less common side effects and potentially a lower risk of myocarditis (albeit, this is low altogether).
Do you envision one vaccine being more readily available?
This all depends on what each clinic/health center orders. They likely will not order both because of cost and storage needs. Moderna does not need to be stored in a low temp freezer (just normal freezer) so may be easier for most practices/health departments. On the other hand, many practices and facilities have been giving Pfizer for children 5+ so are familiar with the mixing and storage. Vaccine storage and administration is expensive and they will likely order one type. There are also many logistical things practices have to consider such as storage and limiting waste given each vial contains 10-12 doses and has to be thrown out after 12-24 hours if not used.
Are you vaccinating Ryaan and which vaccine do you prefer?
To be completely transparent, my husband and I have gone back and forth about whether Ryaan needs to be vaccinated. He has had COVID once (and likely twice) and fared well. My husband is also seeing less and less very sick patients with COVID in his hospital and I’m also seeing less severity now than with delta or alpha. I’m seeing less MIS-C and less long COVID compared to delta.
Hospitalizations and illness are there but severity seems to be declining. So we have asked ourselves..Is this needed?
We ultimately did decide to vaccinate given we cannot predict what future variants will do. But, I share this story to be completely transparent that we are pro-science, vaxxed and boosted adults who work in healthcare, but we also weigh the benefit and risk of everything we put into our son’s or our bodies.
We planned on vaccinating him in August and he had a fever and ear infection the day before his appointment. We are rescheduling for mid-September. He will get Pfizer as that is what’s available to us in our clinic. I also did prefer Pfizer given he has had COVID, we are not in a rush for protection, and it’s a smaller dose.
I also do like that Pfizer has been given to children 5+ whereas Moderna has only been given to adults 18+ so I like the more pediatric data.
Also, although the risk of myocarditis is low overall after COVID vaccines, there is a risk and we want to choose the lowest dose possible that still met immunobridging data (Pfizer). (More to come in another question about myocarditis risk).
Can the vaccine be administered with other childhood vaccines?
Both the AAP and ACIP agree that they can be given with other vaccines. The trials did not directly study co-admin, but there is no reason to be concerned with co-admin per the AAP and ACIP given it has been done with adults with not issue.
I likely will have families who would want to give it alone given it’s more “new” in nature and I would be fine with that. The hope is compliance so if you do it at a separate visit, make sure to return for the series. We will likely get the COVID vaccine solo, unless the flu vaccine is available then he will get both the same day.
Whatever you decide, please make sure to return to complete your vaccine series.
What do I do if my child turns 5 soon. Wait or get series now?
If your child has not had COVID or is immunocompromised, I would get the vaccine as soon as it’s available to you. If your child has had COVID and is immunocompetent, you can wait no more than 3 months. This also depends on when your child turns 5. I would consider vaccination by the fall to account for future variants that may circulate. Remember, there are currently new variants circulating (BA.4. and BA.5) so vaccines could help reduce risk from that.
What do I do if my child starts the series now and turns 5 after starting series? Get the 5-year-old dose or continue with the <5 dose?
In this situation, ACIP recommends getting the dose recommended for the age of your child. So if you got the Pfizer 3mcg and then another 3mcg and then your child turns 5 before the 3rd dose, they would get 10mcg which is the dose for 5+.
Should we wait for closer to school?
If your child is immunocompromised and/or has not had COVID, I would get the vaccine ASAP if you plan on getting it. If your child is not immunocompromised or has had COVID, you can wait until July.
Remember, the full series takes time, so the hope is you have vaccine protection before a future variant surge.
What side effects to watch for?
Common systemic side effects include: fever, fatigue, headache, chills, myalgia, irritability, sleepiness, and/or loss of appetite. If these occur, they should last about 2 days-4 days.
Common local reactions include: pain at injection site, lymph node swelling in underarm (if administered in arm) or groin (if administered in thigh).
The ACIP meeting did stress the importance of reporting side effects to VAERS, especially any outside of these (which we do not expect at all but they want this for full transparency and monitoring).
Should I take a day off after my child gets the vaccine?
The side effects seem to be similar to routine vaccinations children get. I typically always give Ryaan his routine vaccines on Fridays because I do like having the days off after if he has a fever or is irritable, but this is personal preference and not a requirement. Schedules may not always allow this.
Can I give Motrin or Tylenol after vaccination?
Yes. Either are fine at their recommended dosing. Similar to other vaccines, there is no utility in pre-medicating. Give medicine as needed after vaccination for pain/irritability/discomfort.
What about the risk of myocarditis?
The data we have as of now comes from the Pfizer vaccine because that’s the only vaccine that’s been given 5 years+ in the US. As a reminder, children 5-11, received 10mcg and children 12+ receive the adult dose of 30mcg. Through VAERS (where reactions are reported), Boys aged 12-15 had incidence of 46.4/1 million doses of the vaccines. Boys aged 16-17 had an incidence of 75.9/1 million doses. Boys 5-11, had a reported 2.6 cases out of 1 million doses administered. Girls had less of a risk than boys. The orange boxes indicate a slight increase than the typical incidence of myocarditis as it is something that we do see clinically from viruses.
So, there is a risk with vaccination, however this risk is LOW and the benefits of vaccination do outweigh the risk of myocarditis (ex. 46 out of 1 million doses is very, very rare and these children recovered). Neither trial had any children report cases of myocarditis and it will be important for us to monitor data.
There was a discussion during the FDA meeting that Pfizer may have less of a risk of myocarditis given the dose of 3mcg vs 25mcg, however do remember that the risk is overall low and children recovered if they did have vaccine-related myocarditis. Also, this age group may see less myocarditis given children of this age are less likely to have myocarditis than older children as it is.
What are the doses of each vaccine for other age groups?
You can check here for updates on boosters and the schedule (may not be updated by the time I have this sent out!)
Moderna
6 months-5 years: 25mcg
6-11 years: 50mcg
12+: 100mcg
Pfizer
6 months-4 years: 3mcg
5-12 years: 10mcg
>12+: 30mcg
If my child got Moderna as dose one, can they get Pfizer for dose two?
It’s recommended to stick with the series you started, HOWEVER if the original brand is not available you can continue the series with the brand that is available to you.
My baby is premature. Would their dose be smaller?
They would get the recommended dose for their age. We are using chronological age for premature babies (from their birth date) and not adjusted age.
Is the vaccine effective against BA.2/BA.4/BA.5?
Unfortunately, just like for adults we are not certain how these vaccines will respond to future Omicron variants or variants in general. The trial study was done during the initial Omicron surge in January-February.
Will they need this every year like the flu or need boosters?
Unsure at this point similar to adults. We know that this age group will need a minimum 3-dose series like Pfizer has been authorized for and Moderna will likely have a booster by the fall for this age group. How many vaccines besides these are uncertain (similar to adults).
Do you think all kids should get this vaccine?
I completely appreciate concerns over vaccinating children in this age group. They receive many vaccinations as it is and they are (by percentage or hospitalizations/cases) overall lower risk for complications than older children or adults.
I do believe all children should be encouraged to get this vaccine, especially if they have not had COVID yet. However, I do not believe in mandates in this age group given risk by percentage of complications or hospitalizations is low.
It’s important to consider what risk you are comfortable with. 26% of children of this age group that were hospitalized had no underlying medical conditions so we still do not know which children will have a simpler course than others. We also do not know what future variants will do. We can’t predict how this virus will mutate and vaccination can in theory help protect severity of illness against future variants.
The one group I go back and forth between is the group that has had COVID once or twice and has had mild courses. Could this group be okay with future variants? Likely. However, like I already said—we cannot predict how future variants can act and vaccinating prior to new (potentially worse) variants can provide benefits.
There is unfortunately a lot of ‘may” in regards to true efficacy because that’s the same as adults. It’s not the vaccine, but the virus that continues to change. Vaccination is our best bet to reduce risk even futher. I will say that safety trials and real-world safety data in older children have made it that an EUA makes sense, so the choice is yours if you will vaccinate.
Ready to get vaccinated?
- Contact your Pediatrician’s office to see their timeline.
- Go directly to your local pharmacy’s website to see if they are carrying vaccines and scheduling appointments.
- This resource was shared on social media to find vaccines in your area. I have not vetted these resources so please make sure they are legitimate facilities.
- Many pharmacies should be vaccinating children 18 months+ or 3 years + depending on the pharmacy. You can search here for COVID vaccination sites. Sites should be indicating minimum age of vaccination.
- Check out the White House’s press-briefing.
This is a momentous time to be the first country in the world to provide this extra risk-reduction to our children as young as 6 months. As always, I will keep you updated on any further vaccination studies on efficacy and safety with real-world data as they are released.
I hope this blog was helpful. If it was, send it to a friend who may be curious about the COVID-19 vaccines and let’s continue to spread facts about vaccination!
Checkout this podcast episode where I chat with a mom who enrolled her child in a COVID vaccine trial!




